 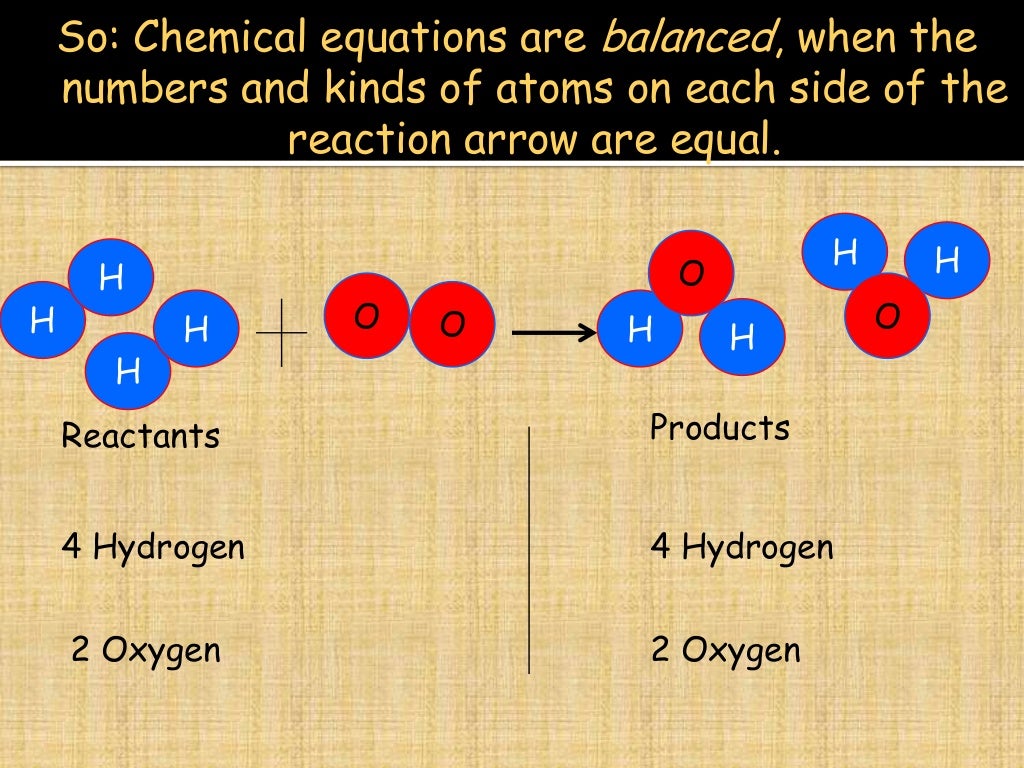
The conservation of mass requires that the sum of the masses of oxygen and hydrogen be equal to the mass of water. The reactants (O 2 and H 2) no longer exist after the reaction and a new product (H 2O) is formed. For example, consider oxidation of hydrogen. Mass is also conserved in chemical reactions. The conservation of mass requires that the mass of water at its initial state (steam) be equal to the mass of liquid water. At the final state, water is in liquid phase. /GettyImages-122688080-5bbb6cd8c9e77c002648731c.jpg)
In this process, water is initially in gas phase, but then it undergoes a condensation process. For example, consider condensation of steam. The total amount of matter in a given process is fixed, but it may change from one form to another. 
In every process, it is necessary to obey the law of mass conservation. This principle like many physics laws is empirical that is, its validity rests on experimental observations. The principle of mass conservation states that matter is neither created nor destroyed. Yousef Haseli, in Entropy Analysis in Thermal Engineering Systems, 2020 1.2 Conservation of mass F oc, organic carbon content NA, not applicable. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
